Business Milestones
![]() has the following go-to-market strategy:
has the following go-to-market strategy:
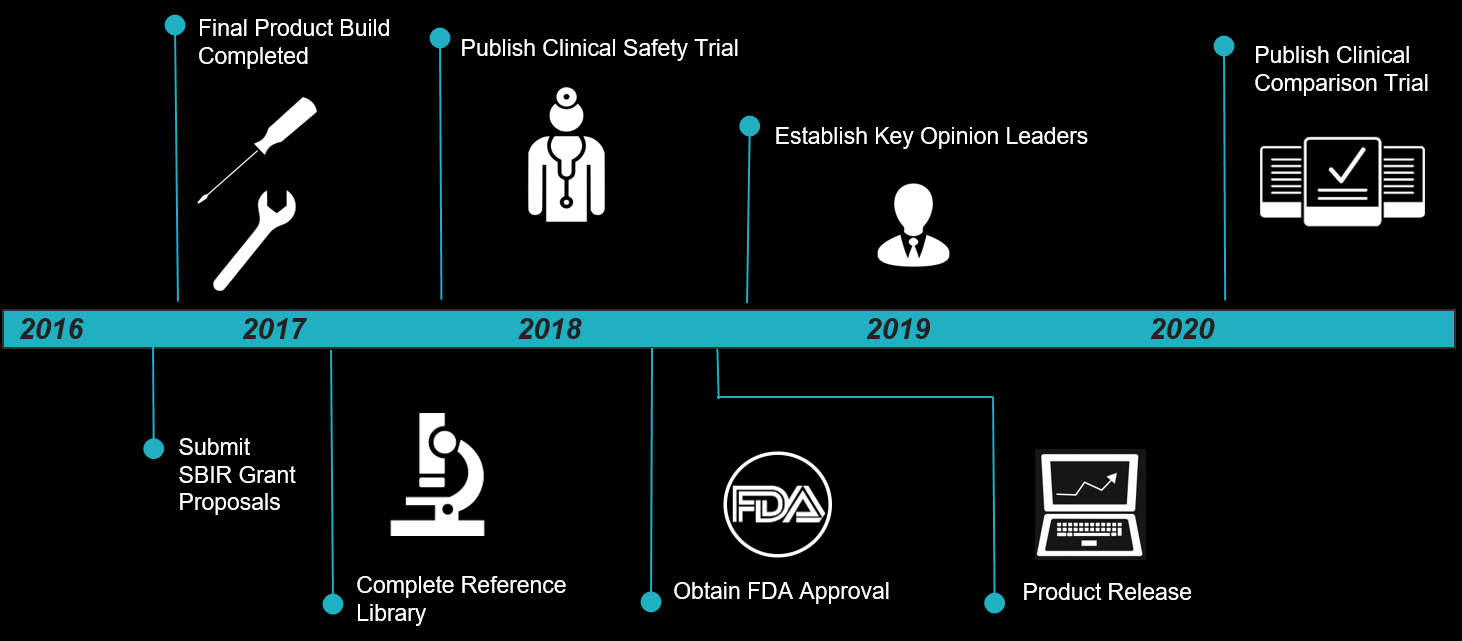
Phase 1: 2016 – 2017
- Prototype Development: Continue to iterate upon our technology while keeping your safety in mind.
- File for SBIR/STTR grant to procure additional funding for phase 1.
- Conduct Animal Studies: Implant mice embryos analyzed by our technology to ensure that embryos approved by
 are indeed healthy and viable. Additionally, continue to build up our reference library of embryos.
are indeed healthy and viable. Additionally, continue to build up our reference library of embryos. - Complete Final Product Build: Finalize the market-iteration of
 which will emphasize miniaturization to seamlessly operate in a clinical setting.
which will emphasize miniaturization to seamlessly operate in a clinical setting. - Complete Reference Library: While the reference library will be completed early, our database will constantly grow from any future measurements.
- Publish Animal Studies: To remain transparent and accountable, we will publish the results to our animal studies.
Phase 2: 2017-2020
- Establish Key Partnership with select IVF Clinics: Partnering with IVF clinics will facilitate the process in conducting our Clinical Safety Trial due to easier access to patients. Additionally, these IVF clinics will benefit in a symbiotic partnership as our technology bolsters their success rates.
- Conduct Clinical Safety Trial on real patients in order to ensure our technology is safe, reliable, and accurate.
- Publish Clinical Safety Trial and Obtain FDA Approval: Again, we will publish the results to our trial for transparency and accountability. The Clinical Safety Trial is critical in obtaining FDA Approval for our device.
- Begin Clinical Comparison Trial comparing our technology alone, our competitors (Embryoscope & Eeva Test), and our technology in tandem with our competitors.
- Establish Key Opinion Leaders (KOLs): Before entering the market, we plan to establish an advisory board of KOLs, experts in the field (embryologists, obstetricians, medical device consultants, business advisors, and more) to give guidance on our operations. Our KOLs will give us credibility while helping to keep you safe.
- Product Release: Launch
 into the market. Any IVF clinics may purchase our technology at this point.
into the market. Any IVF clinics may purchase our technology at this point. - Publish Clinical Comparison Trial: Release the results to our Clinical Comparison Trial to detail how our technology stacks up with the competition.
Key Highlights:
We plan to establish key partnerships with select IVF clinics. These partnerships will facilitate our ability to conduct a Clinical Safety Trial and Clinical Comparison Trial. Our goal is to establish strong and mutually benefical relationships with IVF clinics. These clinics will be able to advertise their higher success rates due to our technology. For us, we will be able to slowly make our name in the IVF landscape.
After we have released our product, we will be pursuing a partnership to couple  as an add-on to the Embryoscope or Eeva Test, with our rationale being that you, the parents, will be the greatest beneficiaries. By combining 2 or more embryo screening technologies, IVF clinics will be able to more accurately select healthy embros.
as an add-on to the Embryoscope or Eeva Test, with our rationale being that you, the parents, will be the greatest beneficiaries. By combining 2 or more embryo screening technologies, IVF clinics will be able to more accurately select healthy embros.
